|
The current model postulates that the core complex is a cargo loading assembly that binds to the cytoplasmic tails of trafficking receptors such as the cation independent mannose-6-phosphate receptor (CI-MPR), Wntless, sortilin and DMT1 via the large VPS35 subunit, ,, ,, ,, ,. Biochemical and genetic studies in yeast and higher eukaryotes have identified two distinct retromer sub-complexes a core trimer composed of VPS35-VPS29-VPS26 (VPS: vacuolar protein sorting) and an associated homo or hetero-dimer of sorting nexin (SNX) proteins, containing combinations of SNX1, SNX2, SNX5 and SNX6. Retromer is a protein complex peripherally associated with endosomal organelles, and controls trafficking of a number of critical cargo molecules within tubulovesicular carriers to the trans Golgi network (TGN),. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.Ĭompeting interests: The authors have declared that no competing interests exist. BMC is supported by an ARC Future Fellowship (FT100100027). 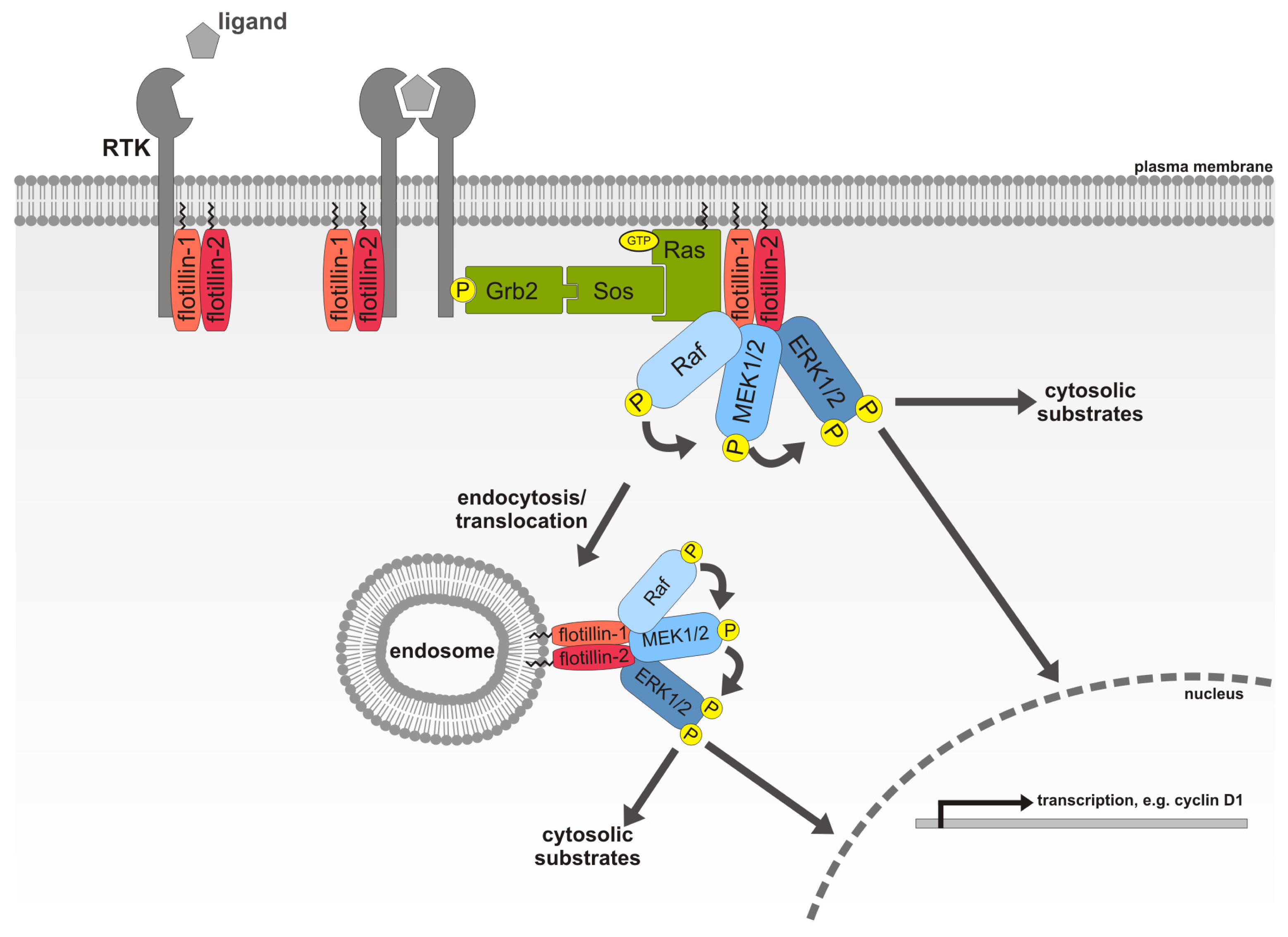
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.įunding: This work is supported by funds from the Australian Research Council (DP0878608), and NHMRC (511072). Received: DecemAccepted: Published: May 24, 2011Ĭopyright: © 2011 Swarbrick et al. PLoS ONE 6(5):Įditor: Ludger Johannes, Institut Curie, France (2011) VPS29 Is Not an Active Metallo-Phosphatase but Is a Rigid Scaffold Required for Retromer Interaction with Accessory Proteins. Our conclusion is that VPS29 is a metal ion-independent, rigid scaffolding domain, which is essential but not sufficient for incorporation of retromer into functional endosomal transport assemblies.Ĭitation: Swarbrick JD, Shaw DJ, Chhabra S, Ghai R, Valkov E, Norwood SJ, et al. Finally, NMR chemical shift mapping indicates that VPS29 is able to associate with SNX1 via a conserved hydrophobic surface, but with a low affinity that suggests additional interactions will be required to stabilise the complex in vivo. There is evidence that structural elements of VPS29 critical for binding the retromer subunit VPS35 may undergo both metal-dependent and independent conformational changes regulating complex formation, however studies using ITC and NMR residual dipolar coupling (RDC) measurements show that this is not the case. We find that although VPS29 can coordinate metal ions Mn 2+ and Zn 2+ in both the putative active site and at other locations, the affinity for metals is low, and lack of activity in phosphatase assays using a putative peptide substrate support the conclusion that VPS29 is not a functional metalloenzyme. In this study we examine the functional interactions of mammalian VPS29, using X-ray crystallography and NMR spectroscopy. VPS29 has a fold related to metal-binding phosphatases and mediates interactions between retromer and other regulatory proteins. 
VPS29 is a key component of the cargo-binding core complex of retromer, a protein assembly with diverse roles in transport of receptors within the endosomal system.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
